 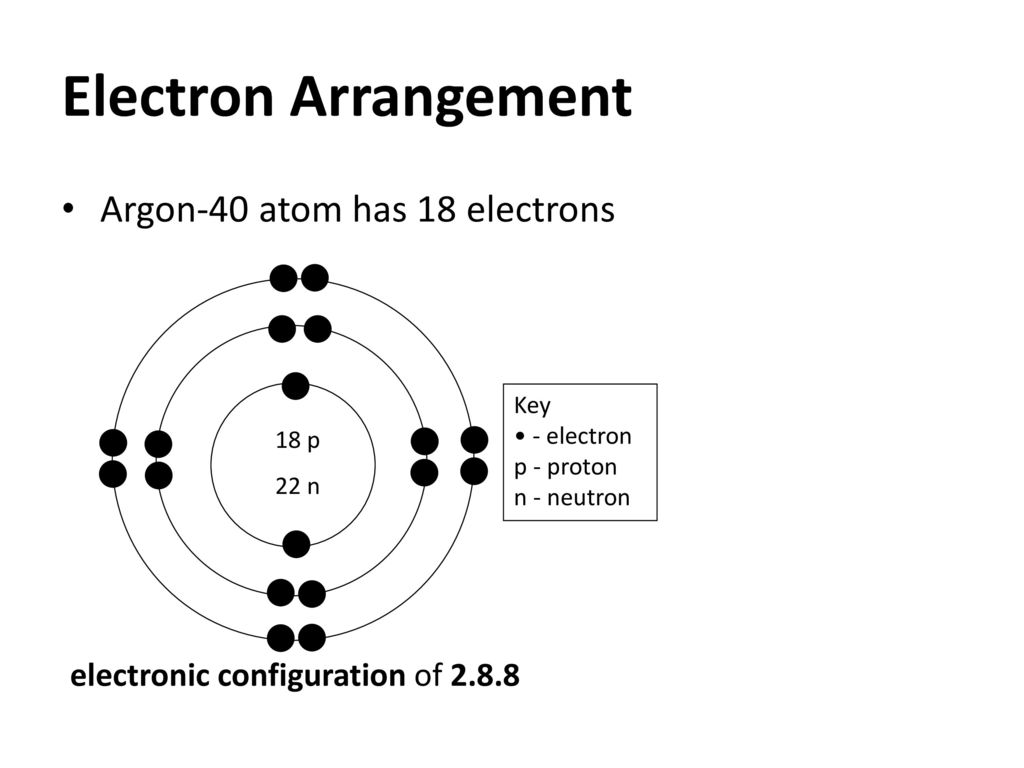
When expressing the same phenomenon as an intensive property, the heat capacity is divided by the amount of substance, mass, or volume, thus the quantity is independent of the size or extent of the sample. Heat capacity C has the unit of energy per degree or energy per kelvin. Heat capacity is an extensive property of matter, meaning it is proportional to the size of the system. When a given amount of heat is added to different substances, their temperatures increase by different amounts. 
Their SI units are J/kg K or J/mol K.ĭifferent substances are affected to different magnitudes by the addition of heat. The properties c vand c p are referred to as specific heats (or heat capacities) because under certain special conditions they relate the temperature change of a system to the amount of energy added by heat transfer. Where the subscripts v and p denote the variables held fixed during differentiation. The intensive properties c v and c p are defined for pure, simple compressible substances as partial derivatives of the internal energy u(T, v) and enthalpy h(T, p), respectively: You might want to place an order with us.Specific heat, or specific heat capacity, is a property related to internal energy that is very important in thermodynamics. Source link "96% of our customers have reported a 90% and above score. The post How do you find the molar mass of argon? appeared first on Essay Conect. Long story short the molar mass of an element can be found by taking its relative atomic mass listed in the periodic table and tagging along the units that correspond to molar mass i.e. Therefore the molar mass of argon which is simply the mass of one mole of atoms of argon is equal to This means that the atomic mass of argon is equal toīy definition a unified atomic mass unit #u# is equal to #1 g mol^(-1)#. You get the relative atomic mass by dividing the atomic mass #m_a# by #u# which means that you can get the atomic mass by multiplying the relative atomic mass by #u# Here comes the cool part - this value is also the element’s molar mass! Here’s why. In this case you have #39.948# which means that the relative atomic mass of argon is Notice the number added to the bottom of the element box. You’ll find it located in period 3 group 18.  
Grab a periodic table and look for argon #Ar#. Argon molecular weight Molar mass of Ar 39.948 g/mol Convert grams Argon to moles or moles Argon to grams Percent composition by element Calculate the molecular weight of a chemical compound Enter a chemical formula: Browse the list of common chemical compounds. An element’s relative atomic mass #A_r# tells you the ratio that exists between the average mass of the atoms that make up an element and #1/12th# of the mass of a single unbound carbon-12 atom i.e. Įvery element listed in has its relative added to the bottom of its element box. The thing to remember about the molar mass of an element is that it’s actually given to you in the Periodic Table of. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
